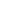Federal PBM Reform - Key Compliance Requirements and Penalties

The regulatory landscape for Pharmacy Benefit Managers (PBMs) has shifted from a period of minimal federal over-sight to one with greater federal involvement amid a patchwork of state laws.
Historically, PBMs have served as third-party administrators responsible for managing prescription drug benefits through claims processing, network design and rebate negotiations. However, the longstanding lack of transparency inherent in these operations has culminated in significant regulatory scrutiny and escalating litigation risks for plan sponsors.
The enactment of the Consolidated Appropriations Act of 2026 (CAA 2026), together with the Department of Labor’s (DOL) proposed fee-disclosure regulations, marks a pivotal transition toward greater federal oversight of PBM business practices. Although both the CAA 2026 and the DOL proposal introduce comprehensive PBM reforms, they differ in scope and timing.
This infographic highlights key requirements plan sponsors need to understand as they prepare for these new federal regulatory developments.
Next Steps
As implementation timelines advance and the DOL moves toward finalizing its rule, plan sponsors and issuers should evaluate their existing PBM contracts and ensure that compliance processes are in place to meet their upcoming reporting, disclosure and fiduciary obligations. Proactive monitoring and early preparation will help ensure that plans remain aligned with evolving regulatory expectations.

 Prev
Prev